|
Today's News |
RepliCel Closes $1 Million Final Tranche of Strategic Investment Commitment and Terminates License Agreement with Shiseido
Tuesday, January 4, 2022Company Profile | Follow Company

Vancouver, BC, January 4, 2022--(T-Net)--RepliCel Life Sciences Inc. (OTCQB: REPCF) (TSXV: RP) (FRA:P6P2), a company developing next-generation technologies in aesthetics and orthopedics, announced that it has closed the final tranche of the investment by MainPointe Pharmaceuticals as outlined in the share purchase agreement signed by the parties earlier this year.
The final tranche involved the issuance of 1,479,882 common shares by RepliCel in exchange for CAD$998,921 which has now been received by the Company. The Company received an aggregate of CAD$2,698,921 from all tranches and issued an aggregate of 3,986,684 Shares at a price of CAD$0.675 per Share.

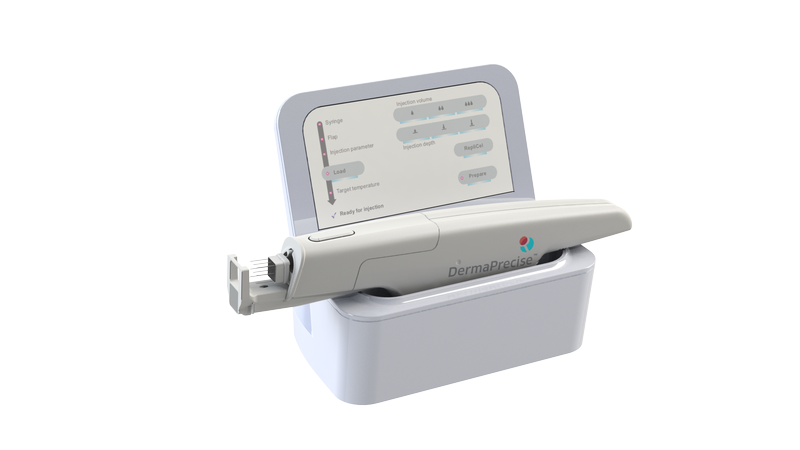
"The timing of this final share purchase by MainPointe," stated RepliCel's President and CEO, R. Lee Buckler, "coincides nicely with the increasing level of collaboration between RepliCel, the European firm conducting the DermaPrecise development, testing, and manufacturing (A.M.I.), and MainPointe's regulatory department committed to handling all aspects of the regulatory submission to the U.S. Food and Drug Administration seeking marketing approval for the injector and related single-use consumables."
RepliCel also announced that the dispute between Shiseido and RepliCel has led to the Company electing to terminate its license agreement with Shiseido. The Company's decision is a legal step in its pursuit of a resolution of its disagreement with Shiseido that is the subject of ongoing arbitration proceedings under the aegis of the International Center for Dispute Resolution (ICDR).
The dispute between Shiseido and RepliCel is related to a license for Asia to RepliCel's cell therapy technology for androgenetic alopecia, which is the leading cause of male and female pattern hair loss. Legal counsel for RepliCel filed a Notice of Arbitration that was announced in a Company press release on October 18, 2021.
About RepliCel Life Sciences
RepliCel is a regenerative medicine company focused on developing cell therapies for aesthetic and orthopedic conditions affecting what the Company believes is approximately one in three people in industrialized nations, including aging/sun-damaged skin, pattern baldness, and chronic tendon degeneration. These conditions, often associated with aging, are caused by a deficit of healthy cells required for normal tissue healing and function. These cell therapy product candidates are based on RepliCel's innovative technology, utilizing cell populations isolated from a patient's healthy hair follicles.
The Company's product pipeline is comprised of RCT-01 for tendon repair, RCS-01 for skin rejuvenation, and RCH-01 for hair restoration. RCH-01 was exclusively licensed in Asia to Shiseido Company (an agreement which is currently in dispute). RepliCel maintains the undisputed rights to RCH-01 for the rest of the world. RCT-01 and RCS-01 are exclusively licensed in Greater China to YOFOTO (China) Health Company. RepliCel and YOFOTO are currently co-developing these products in China. RepliCel maintains the rights to these products outside of Greater China.
RepliCel has also developed a proprietary injection device, RCI-02, and related consumables, which is expected to improve the administration of its cell therapy products and certain other injectables. YOFOTO has exclusively licensed the commercial rights for the RCI-02 device and consumables in Greater China for dermatology applications and is expected to first launch the product in Hong Kong upon it being approved for market launch in either the United States or Europe.
Please visit www.replicel.com for additional information.
Notable Facts:
- RepliCel's three cell therapy products have now been tested in over 100 patients in four countries on three continents having been successfully reviewed by three regulatory agencies.
- RepliCel has key strategic partners in the United States, China, and Japan each of which is now investing heavily in the further clinical testing and development of RepliCel's products for their markets. Data from each of the clinical programs will strengthen the product development initiatives for RepliCel and its other partners worldwide.
All Shares issued are subject to a statutory hold period expiring four months and one day from the date of the issuance of the Shares. None of the securities sold in connection with the investment will be registered under the United States Securities Act of 1933, as amended, and no such securities may be offered or sold in the United States absent registration or an applicable exemption from the registration requirements. This news release shall not constitute an offer to sell or the solicitation of an offer to buy nor shall there be any sale of the securities in any jurisdiction in which such offer, solicitation or sale would be unlawful.
Other Recent Company News  |
|||||||||||||||||||
|
|||||||||||||||||||


 News
News Events
Events Tech Careers
Tech Careers Directory
Directory Tech Stocks
Tech Stocks T-Net 100
T-Net 100 Members
Members Feedback
Feedback About T-Net
About T-Net Employers
Employers Post Job Listings
Post Job Listings Advertise
Advertise